1-Detection of Gases, Gas Concentration Measurements
Before going into the details of LEL and UEL concepts, it is useful to have information about the importance of gas detection systems and gas-related hazards. For more information, you can read article Detection of Gases.
Mass (g, kg) or volume (m3, L) units are used to measure the amount of substances in daily life. The amount of solid and liquid substances is easily indicated in these units, thanks to their mass and volume. While the mass of compressed gases can be determined by measuring the mass of compressed gases in a tube, there is no easy way to measure the volume or mass of gases dispersed in the form of leaks, and even if measured, mass or volume data alone are not useful. We need different measures to specify the amount of gases in the environment.
In this article, it will be explained why we need to measure and what are these measures.
2- What are LEL and UEL?
LEL and UEL are the abbreviations of “Lower Explosive Limit” and “Upper Explosive Limit”, respectively. (Also used as LFL and UFL, which stands for Lower Flammability Limit and Upper Flammability Limit.)
These measurements are useful in specifying the amount of flammable-explosive gases. Flammable gases do not show combustion-explosion characteristics with any amount of leakage, they become flammable in a specific range of combustible gas-air mixture. The lower limit of this range is called LEL, and the upper limit is called UEL. Since the mixture at the ratios below the LEL level is a ‘thin’ mixture in terms of the amount of flammable gas, combustion or explosion will not occur. At rates above the UEL level, combustion or explosion will not occur because there is too much flammable gas in the mixture (insufficient oxygen in the environment).
Each combustible gas has its own LEL and UEL range. LEL and UEL values of some gases are given in the table below.
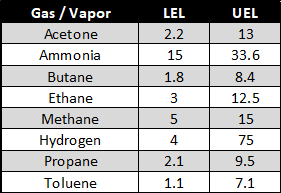
Source: International Labor Organization (ILO)
https://www.ilo.org/dyn/icsc/showcard.listCards3?p_lang=en
3- What is %LEL?
LEL and UEL dimensions indicate the ratio of flammable gas to the mixture of flammable gas and air by volume. To elaborate on the frequently used methane gas example, the air mixture containing 5% methane is flammable-explosive. Even when the methane content of the mixture is increased up to 15%, it remains flammable. A mixture outside this range is not flammable or explosive. As can be seen, the amount where the dangerous situation starts in flammable gases is the point where the LEL level of the leaking flammable gas is reached. This is the reason why the concept of LEL appears more often than the concept of UEL in general usage.
In gas detection, it is a useful way of specifying the amount to know how close the LEL value is, rather than knowing the proportion of flammable gas in the mixture of flammable gas with air. The unit of measurement used when specifying this amount is %LEL. 2.5% by volume of methane gas in the environment can be specified as 50% LEL. 5% methane indicates that the 100% LEL level has been reached and there is a risk of explosion.
4- What is %vol?
While mentioning about the dangerous gases, it was explained that it does not make much sense to know the mass or volume of the gas that has spread to the environment. The typical dangers associated with gases are suffocation, toxicity and flammability. To explain each danger respectively, suffocating gases become dangerous by reducing the oxygen ratio of the air in the environment to an insufficient level, toxic gases become dangerous by mixing with the air we breathe in a small amount, and flammable gases become dangerous by forming a flammable gas + air mixture depending on the flammability level of the gas.
Considering the common point here, the proportion of the dangerous gas in the air gives a clue about the danger. Therefore, the most basic way of specifying the amount is vol. % (volume percent). Since the volume of gases varies greatly under pressure and temperature, unless otherwise specified in vol. %, volume ratios are specified based on the gas mixture at 20°C temperature and sea level air pressure. (15°C – 25°C in some sources.)
For example, when talking about the gases in the earth’s atmosphere, the volume ratios of the gases in the lowest layer, the troposphere, are given, such as 78.09% nitrogen, 20.95% oxygen, 0.93% argon. In the upper layers, these mixing ratios also change with pressure and temperature.
The vol. % measure is useful for describing ambient oxygen and suffocating gases. The human body sustains its life by breathing in an atmosphere-air mixture containing approximately 21% oxygen. Even gases that do not have any toxic effect, when leaking into the environment in high quantities, can create a suffocating gas effect by forming an air mixture with insufficient oxygen ratio, which is not suitable for breathing. According to US Department of Labor data; air with up to 19.5% oxygen is safe to breathe, while oxygen levels below 19.5% are considered “oxygen deficient” and are outside the safe range. (source: https://www.osha.gov/laws-regs/standardinterpretations/2007-04-02-0)
5- What is PPM and PPB?
It is an abbreviation for “parts per million”. Similar to the percentage (%) that is frequently used, the ppm measure is not a dimensional measurement unit like kg or L, but a dimensionless measure that indicates the ratio of substances in mixtures to the whole mixture. For example, it is used to indicate a proportional measure not as “there is 1 liter of gas in the air”, but as “one millionth of the air consists of this gas”.
In gas detection, this form of measurement is most useful when measuring and expressing the amount of toxic gases. It is mentioned that flammable-explosive gases start to show flammable-explosive properties (LEL) when the volumes of the related gases in the environment vary between 1% ~ 15%. For toxic gases, percentage expressions are insufficient, and gas amounts at much lower rates gain importance. Even very little presence of some gases in the environment can cause poisoning. For example, ammonia from common toxic gases at 25 ppm, carbon monoxide at 35 ppm, and hydrogen sulfide at 5 ppm begins to pose a danger to human health. (It can be thought of as 25/35/5 out of a million particles in the air.) Toxic gas detectors should be used for personnel safety in potential areas where these toxic gases can be found, and the gas concentration should be continuously monitored by measuring at the ppm level.
For some toxic gases such as formaldehyde or chlorine, this limit is much lower, such as 0.016 ppm or 0.1 ppm. For highly toxic gases like these, a smaller unit of measure than the PPM measure, PPB, or “parts per billion” can be used. In working environments where toxic gases may present, the permissible concentration of the relevant gas is determined by international organizations. For example, there are limits such as the average exposure limit in a 10-hour working day, the exposure peak value that should not be exceeded at any time of operation, the short-term exposure value that should not be exceeded in a short interval of 15 minutes, the immediate danger to life or health limit that requires immediate leaving the environment. These toxic gas exposure limits are usually indicated by the PPM measure.
6- What is mg/m3?
Another unit of measurement frequently used in toxic gas concentrations is mg/m3. The concepts we have seen so far are representations as the ratio of the volume of the target gas to the volume of the gas mixture it forms with air. (Volume/Volume)
Mg/m3 unit is the ratio of the mass of the target gas to the volume of the gas mixture formed with the air (Mass/Volume). In other words, it is a form of measurement that indicates how many milligrams of the target gas are present in 1 cubic meter of air. Based on 25°C temperature and 1 atm pressure unless otherwise stated.
The mg/m3 measure is a form of measure that indicates the mass/volume ratio. Therefore, it is necessary to know the molar mass of the relevant gas and the volume constant of a gas under the relevant temperature-pressure when converting from mg/m3 to other measurement units or when converting other measurement units to mg/m3. Since it is not practical to calculate, the ppm value is usually given together with the mg/m3 value for toxic gases.
7- How to make %LEL – %VOL – PPM Conversions?
It was mentioned that percentages and million ratios are used in vol. % and PPM measurement units. Accordingly, we can calculate 1 percent as equals to 10 thousand per million.
1% vol = 10,000 ppm.
0.1% vol = 1,000 ppm
0.01% vol = 100 ppm
0.001% vol = 10 ppm
0.0001% vol = 1 ppm
The % LEL value can be used with the gas name as each gas has its own variable LEL value.
If we look it on the example of hydrogen sulfide (H2S), which is both toxic and flammable gas:
100% LEL = 4% vol = 40,000 ppm H2S
50% LEL = 2% vol = 20,000 ppm H2S
10% LEL = 0.4% vol = 4,000 ppm H2S
Please do not copy or cite our content without permission. Feel free to reach out for authorization.
Karf&Scoot










